|
Mn increases strength and wear resistance of the steels. Įven small concentrations (1.5%) of copper are capable of increasing the resistance to reducing acids (e.g. N can be used to achieve high surface hardness due to the formation of nitrates during nitrating.Ĭa increases, even in small amounts and under certain smelting preconditions, the mechanical workability without negatively influencing the general performance properties. As an alloying element N stabilises the austenite structure especially and increases in austenite steels the strength, the yield strength as well as the mechanical properties when subject to heat. N can both damage steel as well as be used as an alloying element. So-called carbide formers are frequently found in corrosion resistant steels Ti has a stabilising effect on inter-cyrstalline corrosion. Ti has a highly deoxidising, denitrating effect, binds sulphur and forms carbide. Mo is one of the elements that increases corrosion resistance and is therefore frequently added to highly alloyed Cr steels and Austenite Cr-Ni steels. Mo also reduces the annealing brittleness of Cr-Ni steels and improves weldability. Moreover, machining steel produces shorter swarf during machining.Īlong with other elements, Mo increases the hardenability of the steel. The lubricant effect on the tool cutting edge and as a consequence the reduced friction between workpiece and tool lengthen tool life. Si and Al create ferrite they increase the scaling resistance (scaling temperature) especially of ferrite steels (chromium steels with a relatively low carbon content).Īlthough this tends to have a deleterious effect on steel, up to 0.4% S is deliberately added to machining steel. Mn increases strength and wear resistance of steels. 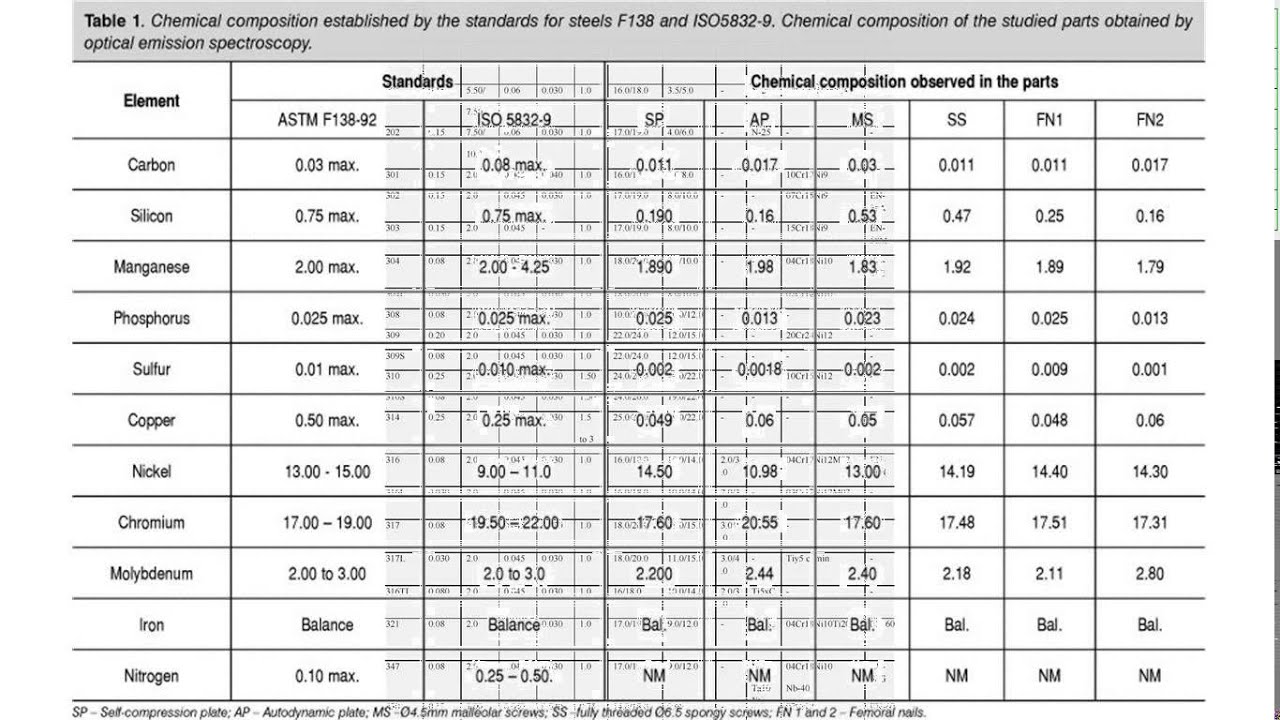
Cr-Ni steels are practically impossible to magnetise and are characterised by low heat and electrical conductivity. Moreover, Ni in austenite Cr-Ni steels enhances resistance to the influence of reducing chemicals. In percentages of more than 7%, Ni added to a high Cr content produces chemically robust steels exhibiting an austenite structure until well below room temperature. A Cr content of at least approximately 13% is required for the corrosion resistance of the steel. The tensile strength of the steel is increased by adding Cr. Likewise, the more Cr in the steel, the worse its weldability. It does, however, reduce the notch impact strength as well as ductility. As the carbon content increases, so too does the strength and the hardness of the steel, whereby its tensile strength, forgeability, weldability and workability are reduced.Ĭr improves the hardness and thus the hardening and tempering characteristics of steel. The addition of additional alloying elements to achieve specific properties produces alloyed steel. Structure Standard EN (Europe) No AISI C max Cr Ni Mn max P max S max Si max Mo Other elements A: X10 CrNi 18-8. Chemical composition of stainless steels - Inox. 
Carbon is the most important and influential alloying element in steel. Main features of the most important stainless steels used in food and processing industries Chemical composition of stainless steels (inox).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
